 
Where m is the mass, v is the velocity, r is the radius of the orbit, h is Planck's constant and n is a positive integer. These states of allowed electronic motion are those in which the angular momentum of an electron is an integral multiple of h/2 π or one can say that the angular momentum of an electron is quantized. This higher energy but less stable shell or level is termed as the excited state of the electron. It may move to a level of higher energy or a less stable shell by absorbing energy. The energy level occupied by an electron usually is called the ground state. As one moves away from the nucleus, the energy of the levels increases. The necessary centripetal force is provided by the electrostatic attraction of the protons in the nucleus. In each of these energy levels, electrons move in circular orbit around the positive nucleus. Each energy level has a definite energy associated with it. The electron in an atom has only certain definite stationary states of motion allowed to it, called as energy levels. The Bohr model shows that the electrons in atoms are in orbits of differing energy around the nucleus. We discuss the various postulates of the Bohr Atomic Model. Neil Bohr explained the line spectrum while developing the model of an atom. He also introduced the correspondence principle which states that the spectrum is continuous and the frequency of light emitted equals the frequency of the electron. Absorption occurred when a transition occurred from a lower stationary state to a higher stationary state. The difference between the energies of the two states was radiated as a single photon. Radiation occurred only when an electron made a transition from one stationary state to another. This was a hypothesis, but at least a working one. We discuss the postulates of Bohr Atomic theory.īohr introduced the concept of radiation less orbits in which the electrons revolve as usual around the nucleus but without radiating any kind of energy which is contrary to the laws of electromagnetism. Various other concepts like radiation less orbits and stationary states were also introduced in the Bohr Model. It retained some of the features like the orbiting of nucleus and the electrons under the action of the Coulomb’s Law of electrostatic attraction. Concepts of Physics by HC Verma for JEEīohr Model was introduced after the Rutherford’s model. 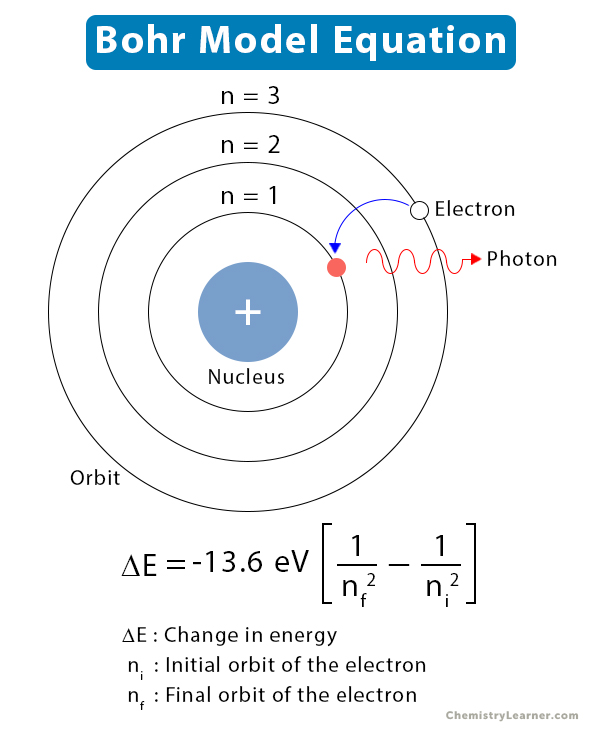
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
